
Furthermore, streamline evolution and vortex emergence under a high Reynolds number 31, 32, electrostatic effects within the buffer solution at different ionic concentrations 33, and shear-thinning and elastic effects in non-Newtonian fluids 34 have been used to actively modulate the D c of the device. Electric field coupling has also been investigated 19, 27, 28, 29, 30. One approach is to actively change the device geometry by stretching the elastic device 24 or changing the flow direction with respect to the pillar array 9, 25, 26. Recently, novel approaches to tune D c actively have been developed. Thus, there has been a significant demand for methods capable of modulating the D c actively to allow using the same DLD device for multiple applications under optimized conditions. Also, fouling and non-specific adsorption on the channel surface can unwantedly change the D c over time, degrading the separation performance of the device. The fixed D c can cause complications when processing biological particles, whose sizes vary considerably by the exact experimental conditions. This is because the critical diameter ( D c), which determines the migration of a particle of a particular size through the pillar array, is generally fixed by the device geometry. To date, the DLD technology has been proven promising for the separation of various particles, including synthetic beads in the nanometer to millimeter range 4, 7, 8, 9, water-in-oil and oil-in-water emulsion droplets 10, 11, and biological particles in the nanometer (e.g., DNA 4, 12 and exosomes 7) to micrometer (e.g., blood cell subtypes 13, 14, 15, 16, circulating tumor cells 17, 18, 19, 20, 21, 22 and viable mammalian cells 23) range.Ī drawback of traditional DLD is that each DLD array can be effective only for a narrow range of predetermined particle sizes. For example, deterministic lateral displacement (DLD) 4 is a promising technology that allows for the continuous separation of suspended particles based on their effective sizes at a high resolution through the pillar array 5, 6. Currently, various microfluidic technologies for particle separation are attracting attention because they can minimize the volumes of precious samples and reagents as well as integrate other elements for comprehensive analysis 1, 2, 3. Separation and purification of the suspended particles of interest are crucial for the sample-preparation steps in analytical chemistry. Further, we perform on/off operation of the particle separation (7-μm and 2-μm beads) by adjusting the D c values. Using the PNIPAM pillars confined in a poly(dimethylsiloxane) microchannel, we demonstrate continuous switching of particle (7-μm beads) trajectories (displacement or zigzag mode) by adjusting the D c through temperature control of the device on a Peltier element. Upon heating and cooling, the PNIPAM pillars in the aqueous solution shrink and swell because of their hydrophobic-hydrophilic phase transitions as the temperature varies. Here, we propose a novel DLD that uses the pillars of a thermo-responsive hydrogel, poly( N-isopropylacrylamide) (PNIPAM) to flexibly tune the D c value. In conventional DLD, the critical diameter ( D c), which determines the migration mode of a particle of a particular size, is fixed by the device geometry. E) the number of protons.Deterministic lateral displacement (DLD) is a promising technology that allows for the continuous and the size-based separation of suspended particles at a high resolution through periodically arrayed micropillars. B) the mass number C) the mass number + the atomic number. 27) The number of neutrons in an atom is equal to A) the atomic number. E) The A groups contain the transition elements. C) The elements in each group have similar chemical properties D) The B groups contain the representative elements. B) A period is a column on the periodic table. 25) What is the correct symbol for the element mercury? A) Mr B) Na C) Me D) Hg E) Au 26) Which of the following is a characteristic of the modern periodic table? A) A group is a horizontal row on the periodic table. E) very large relative to the size of the molecules. D) small relative to the size of the molecules. C) fixed relative to the size of the molecules. B) close relative to the size of the molecules. 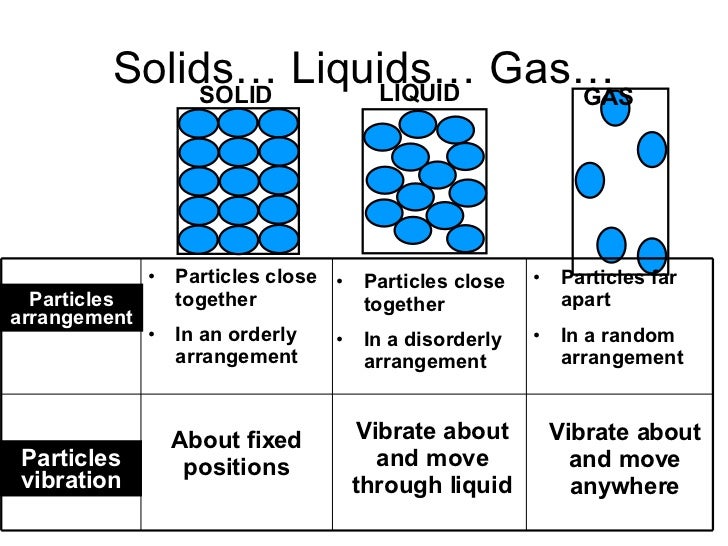 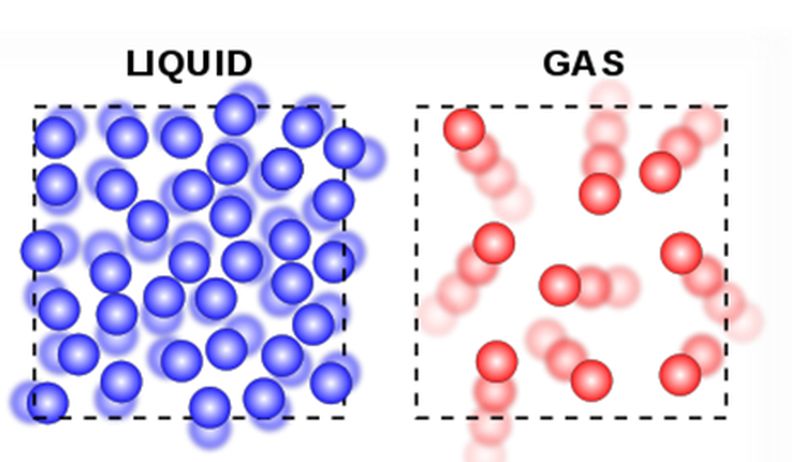
24) In a gas, the distance between the particles is A) very close relative to the size of the molecules.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
